India's Cosmetic Regulations 2020
Figure 1- Drug & Cosmetic Act 1940 and Cosmetic Rule 2020
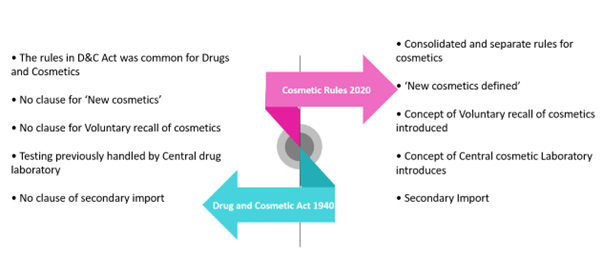
The Drugs and Cosmetics Act 1940 and the Drug and Cosmetic Rules 1945 have regulated cosmetic products in India for years. According to the Drugs and Cosmetics Act 1940, a cosmetic is defined as “any article intended to be rubbed, poured, sprinkled or sprayed on, or introduced into, or otherwise applied to, the human body or any part thereof for cleansing, beautifying, promoting attractiveness or altering the appearance, and includes any article intended for use as a component of cosmetic”.
On 15 December 2020, the Ministry of Health and Family Welfare published the Cosmetics Rules, 2020 in the Gazette of India, under the provisions of the Drugs and Cosmetics Act 1940. The Rules entered into force on the date of their publication in the Gazette, replacing the Drug and Cosmetics Rules 1945.
The Cosmetics Rules, 2020 lay down the regulatory requirements for the manufacture, testing, labelling, import, registration, distribution, and sale of cosmetics in India. The rules consist of 72 rules, 13 schedules and 24 appendixes.
The concept of “new cosmetic” was introduced for the first time. According to the new regulation, the term ‘new cosmetic´ refers to “cosmetic which contains novel ingredient and has not been used anywhere in the world or is not recognized for use in cosmetics in any National and International literature”.
· A prior permission from the Central Licensing Authority (CLA) and submission of data about the products’ safety and efficacy is required when manufacturing or importing a new cosmetic.
· The Cosmetic Rules 2020 has simplified application, reducing the duplication of regulatory processes and formalities for identical products and brands.
· Compliance with Good Manufacturing Practices (GMP) and other guidelines need to be self-declared by the applicant of the registration.
· Products manufactured outside India need to comply with the Cosmetics Rules 2020 (formulation, label, and claims) and need to be registered through the e-Governance portal (SUGAM).
· If the manufacture, sale, or distribution of a product is prohibited in the country of origin, it cannot be imported to India.
How can GCRS help?
At GCRS, maintaining and verifying the information ensuring that the product formulation is safe for consumption in the market, managing the volume of data and ensuring every relevant regulatory requirement is completed before the product goes in the market.
Our representatives have an experience of more than 15 years of working in the industry and will ensure compliance of your overseas and domestic cosmetics and personal care products when needed.
Our team brings essential knowledge and expertise to your brand which end-users expect.
If you have any further questions about the Indian cosmetics compliance, or other regulatory questions. Please feel free to contact us.
GCRS Global









